Other compounds
Lactones
In fresh butter, precursors to lactones and free lactones exist in small concentrations. Free lactones exist in the lipid phase of butter, where they have higher threshold values. Despite their low concentration in fresh butter, free lactones are important flavorants, which act in an additive manner to impart the perceptible sweet, fruity flavors characteristic of butter. Upon heating, the lactone precursors are converted to lactones and their total concentration rises above their FTV. Thus, they provide the rich flavor notes commonly associated with heated foods containing butterfat. Lactones in butter are also the major source of flavor in confections and high-quality candies where they provide the unique, pleasurable flavors associated with these products.
Dimethyl Sulfide
Dimethyl sulfide is originally derived from the feed of cows and occurs in butter at concentrations above its FTV. Dimethyl sulfide helps to smooth the harsher flavor notes of diacetyl and other acidic substances in butter and is also largely responsible for the freshly cooked note associated with freshly churned butter.
Other flavor constituents
Although the exact role of aliphatic aldehydes in butter flavor has not been defined, it is known that they are important. Aldehydes are chemical compounds of the general formula R-CHO (where R is an alkyloran aryl group). They are typically present in concentrations below their Flavor Threshold Value (FTV), particularly since they have low thresholds and produce desirable creamy, buttery flavor at very low concentrations. At high concentrations, they lead to the oxidized off-flavors associated with butter oxidation. Aldehydes can also be found in butter cultures used in the manufacture of cultured butter.
Indole and skatole are two additional flavor compounds which are present in butter and which contribute to its flavor. Phenol and cresol are of borderline significance but also play a role in the flavor of butter.
Additives
BHA (2-Tert-Butyl-4-Methoxyphenol C11 H16 O2)
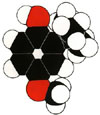 The shelf life of margarine and other fats and oils is improved if oxygen attack on the double bonds in the carbon chains can be repelled or at least diverted, for then they do not turn rancid. The compound 2-tert-butyl-4-methoxyphenol or, more succinctly, BHA is anantioxidant, a substance that inhibits oxidation; it acts by interupting the chain reaction in which oxygen combines with double bonds and slices molecules in two. It does so by combining with peroxide radicals (radicals of the form X-O-O, where X is th rest of the molecule) before they have time to attack other molecules and continue the chain.
The shelf life of margarine and other fats and oils is improved if oxygen attack on the double bonds in the carbon chains can be repelled or at least diverted, for then they do not turn rancid. The compound 2-tert-butyl-4-methoxyphenol or, more succinctly, BHA is anantioxidant, a substance that inhibits oxidation; it acts by interupting the chain reaction in which oxygen combines with double bonds and slices molecules in two. It does so by combining with peroxide radicals (radicals of the form X-O-O, where X is th rest of the molecule) before they have time to attack other molecules and continue the chain.
Antioxidants have been used in foods for thousands of years, but their rules has only recently been appreciated. Among the more familiar ones are spices, which not only mask unpleasant odors but also help to prevent their formation. Sage, cloves, rosemary, and thyme all contain phenolic compounds resemblinh BHA, which interupt the chain reactions and stabilize fats against oxidation. Thyme oil is also effective against bacteria and has been used in gargles, mouthwashes, and disinfectants. Animals contain antioxidant materials, including vitamin E, that serve a similar purpose - to stop them from going rancid while they are still alive.
