|
The Vinland Map: Still a Forgery
Kenneth M. Towe
Department of Paleobiology, Smithsonian Institution, Washington, D.C. 20560
Published in Accounts of Chemical Research, November 1990.
[Dr. Towe retired as Senior Scientist in 1996]
A complete chemical and microscopical reexamination of the Vinland Map resulted in the presentation of new evidence1 regarding this controversial document. It has been concluded that this new evidence "...argues strongly against the specific McCrone Associates proof that the Map is fraudulent". I argue here that this reexamination did not invalidate the basic McCrone Associates conclusion and, to the contrary, further supports their original interpretation of the document as a modern forgery.
Introduction
In 1965, one day before the annual celebration of Columbus Day, the Vinland Map was brought to the attention of the general public.2 With an imputed relevance to the Norse discovery of America, the Map generated much publicity and controversy.3 Nine years later, in 1974, Walter McCrone Associates, Inc., reported4 the presence of titanium dioxide (anatase) in small fragments of ink carefully removed from the Map. Because titanium dioxide was not a pigment available until the 20th century, the McCrone team concluded in their report to the Yale University Library that the Map is a modern forgery. They presented their data later that year as part of a specially convened symposium on the Vinland Map held at the Royal Geographical Society in London.3
However, 13 years after the McCrone report, interest and concern over the Map's provenance and authenticity remained. On the basis of an extensive and nondestructive, "global" microchemical reinvestigation of the entire Vinland Map, in 1987 Cahill et al.1 reported that they were able to find very little titanium in the inks of this supposed 15th century document. Without claiming authenticity for the Map, they nevertheless seriously challenged the 1974 McCrone Associates interpretation and thereby reopened the question of the Map as a fraudulent document. Notwithstanding a rebuttal by McCrone,5 the compositional and microscopical evidence presented by Cahill et al., as well as the conclusions drawn therefrom, deserves additional discussion and interpretation.
Compositional Evidence
In reopening the question of the authenticity of the Vinland Map, Cahill et a1.1 chose to use a technique unavailable to forensic research in the early 1970s: the PIXE (proton-induced X-ray emission) milliprobe. In doing so, the Cahill team placed their analytical emphasis on a comprehensive search of the Map's inks and parchment for titanium, recognizing that it was the finding of this crucial element that raised McCrone's suspicions about the Map in the first place.4 It is important, however, to reemphasize here that it is not elemental titanium alone that is significant to a forgery interpretation. The overall properties of the specific titanium dioxide mineral, anatase, and its distribution on the document are the critical and definitive issues.
McCrone Associates identified, characterized, and illustrated anatase from microparticle samples which they carefully isolated from the Map's inks.3-5 They found that this anatase occurs inhomogeneously dispersed in the ink in a well-crystallized form. The anatase crystals have a rounded rather than angular particle morphology, and they exhibit a narrow particle-size distribution.1,6
Anatase particles having all of these properties can only have been derived from a 20th century source because of the several important and sequential steps that are required in their formation. Firstly, a TiO2 precipitate must be prepared by hydrolysis. Observed in the transmission electron microscope, samples of this initial precipitate are seen to be an aggregated mixture of poorly crystalline to amorphous, very finely divided (~50 Å) anatase "hydrate" crystallites (Figure 1). Secondly, the achievement of both the high crystallinity and rounded crystallite morphology requires that the precipitate pass through a calcination stage. The reaction kinetics of this calcination process demand temperatures in the range of 600-900 °C.7 Finally, to achieve the particle dispersion and narrow particle size distribution desired in commercial pigments and observed in the Map's inks (Figure 2), this calcined product must be appropriately milled to break down and disperse the aggregates of rounded crystals (compare Figure 3 with Figure 2). Given the three stages involved, it is difficult to argue that any such sequential process would have been available (intentionally or accidentally) in the 15th century which would produce an ink with anatase particles having all of the attributes of a modern paint pigment.
Poorly crystalline anatase aggregates with a particle-size distribution overlapping that found in the Vinland Map's inks have been prepared under plausible 15th century conditions.8,9 However, these preparations display broadened X-ray diffraction maxima for anatase, indicating poor crystallinity and/or very fine crystallite sizes. Transmission electron microscope study of such simulated preparations reveals that the particles are aggregates, each made up of numerous fine crystallites, characteristics that compare most favorably to the initial precipitate stage of modern pigment preparations (compare Figure 4 with Figure 1). Therefore, while the simulated pigment is mineralogically anatase, it is not the same as the anatase seen in the inks from the Map. The differences are due primarily to the absence of a calcination step in the simulation experiments and secondarily to the absence of the milling procedure.
|
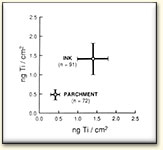
Figure 5. Statistical separation at the 95% confidence level in the mean values of the ink vs the parchment for all of the PIXE titanium determinations made on the Vinland Map. Data from Appendix A, Cahill et al.

|
Cahill et al. do not address either the issue of mineralogy or the crystallography, preferring to emphasize the "global" nature of their comprehensive search and their calculation of the low absolute amounts of elemental titanium on the Map. Provided by Dr. Cahill with a courtesy copy of his original report" to the Yale University Library, I undertook a statistical analysis of all of the PIXE data for titanium as given in Appendix A. This analysis reveals that there is a 95% statistically significant, positive correlation between titanium content and the 91 ink samples measured on the Map without overlap from the 72 titanium measurements made on the parchment (Figure 5). The titanium is therefore clearly enriched in the ink relative to the parchment over the entire Map. McCrone Associates made a similar observation, failing to find any anatase crystals in any of their samples taken from the parchment.4
With titanium enriched in the ink relative to the parchment, it is significant that, in their microanalysis, McCrone Associates obtained a clearly identifiable Debye-Scherrer X-ray powder diffraction pattern for anatase (and calcite) from micron-sized ink fragments. The X-ray film is clear, and the lines for anatase are uniform and sharp. The sharpness of the lines is consistent with a mean crystallite size greater than about 1000 Å. The X-ray films lack evidence of spottiness (except for those lines attributable to calcite).8 The uniform anatase lines imply that there were numerous titanium dioxide crystals present in the fragments. This is in contrast to the electron diffraction films, where selected area analysis means that fewer crystals contribute to the pattern. The nonuniform, spotty lines on the X-ray films indicate the presence of a few large calcite crystals mixed in. The absence of such spots superimposed on the uniform anatase lines, especially the stronger lines, indicates that no additional larger crystals of anatase were present. A wide range in crystal size for anatase is therefore precluded.
All of the major diffraction maxima expected from anatase are present on the film.5,8 A sharp, complete X-ray powder diffraction pattern requires that a substance be well-crystallized and present in quantities above about 0.2-1%.11,12 No such X-ray pattern would therefore have been possible from any fragment having the maximum titanium value of "10 ng/cm2, or 0.0062% by weight" as reported by the Cahill group.1,10
There is thus a clear disparity between the different approaches and their results. With titanium enriched in the ink relative to the parchment and given the lower, millimeter‑level spatial resolution of the PIXE technique, the weight of these results favors the McCrone microparticle analysis.
Microscopical Evidence
As a possible explanation for the microscopic characteristics of the Vinland Map ink lines, McCrone Associates3-5 offered the suggestion that a forger very skillfully applied a double application of ink, carefully putting down a black line over the top of an earlier applied brownish-yellow line. In support of the skilled forger hypothesis is the fact that, to date, only one discrepancy between the yellow and black lines has been observed. The sole error appears on the Map at the western coast of Britain.8 Cahill et al.1 have reconfirmed this characteristic accuracy of the Map and have documented the precise nature of this single discrepancy with high-magnification color photography (plate 5, ref 10).
In describing the other inked lines, they went on to state: "We observe excellent register throughout the Map even in the extremely fine inscriptions, typically to about 0.1 mm. The original edges of the lines can be seen clearly in nearly all parts of the Map and even in the more faded yellowish brown sections of the lines small residual black particles usually extend to the clearly defined limit of the original edge."
For comparative purposes, Cahill et al.1 examined "...many 15th and 16th century vellum manuscripts of undoubted authenticity at the Bancroft Library at the University of California, Berkeley". In general, these documents looked to them very much like the Vinland Map. They observed flaking black layers of pigment over brownish-yellow vestiges. However, in detail, and in marked distinction to their description of the Vinland Map, they report "...places where parts of brownish yellow lines are out of the registration completely with the blacker vestiges of lines". They also noted "...many places in these documents where the brownish yellow vestigal lines appears [sic] alone, without any trace of black on them"; again a marked contrast to their description of the Vinland Map.
Thus, although the Vinland Map is generally similar to the other documents studied microscopically, in detail the characteristics of the Vinland Map ink are quite different.
Discussion and Conclusion
If one accepts the validity of the new microscopical observations made by Cahill et al. on the Vinland Map and the many other indisputably genuine documents studied, it is difficult to understand how the Vinland Map, if it was also genuine, could be so good, with ink so "well-preserved" that it is distinctly different from all the others.
Why are its yellow and black lines not like the genuine documents and completely out of registration in more than one place?
Why are its yellow lines not like the genuine documents and also devoid of any black particles in many places?
The clear disparity between the microscopical descriptions made of the Vinland Map and those of the indisputably genuine documents must be explained in any argument that the Map is itself a genuine 15th century document. Unless plausible answers to these questions can be given, the microscopical evidence gathered by Cahill et al. from the other undoubtedly genuine documents, rather than detracting from the McCrone interpretation, must add additional weight to the conclusion that the Vinland Map is a painstakingly clever forgery.
The microchemical evidence is equally difficult to place in context with the Map's authenticity:
How could a genuine 15th century document have been drawn with an ink containing particles that are mineralogically and crystallographically indistinguishable from a modern paint pigment whose manufacture requires specific and sequential technological procedures?
If pigment-quality anatase had been unknowingly or accidentally added to the Map's surface at some later date, how could titanium be enriched on the inked lines and lettering throughout the Map, but not appear, above background, on the parchment itself?
With all of the chemical, microscopical, historical, and cartographic data now available, it is increasingly difficult to argue that the Vinland Map could still be an authentic 15th century document. If further research is to be devoted to this document and its provenance, perhaps it should be addressed toward an attempt to identify the forger(s), the source(s) of the inks, and the motives for its forgery.
I thank T. A. Cahill, K. D. Harrington, W. A. Kampfer, Walter C. McCrone, Lucy McCrone, and J. S. Olin. The referees provided useful suggestions for improvement of the manuscript.
References:
- Cahill, T. A.; Schwab, R. N.; Kusko, B. H.; Eldred, R. A. Möller, G.; Dutschke, D.; Wick, D. L.; Pooley, A. S. Anal. Chem. 1987, 59, 829‑833.
- Skelton, R. A.; Marston, T. E.; Painter, G. D. The Vinland Map and the Tartar Relation; Yale University: New Haven, CT, 1965.
- Wallis, H.; Maddison, F. R.; Painter, G. D.; Quinn, D. B.; Perkins, R. M.; Crone, G. R.; Baynes-Cope, A. D.; McCrone, W. C.; McCrone, L. B. Geogr. J. 1974, 140, 183‑214.
- McCrone, W. C. Chemical Analytical Study of the Vinland Map; Report to Yale University: New Haven, CT, 1974.
- McCrone, W. C. Anal. Chem. 1988, 60, 1009‑1018.
- McCrone, W. C. Morphology of Ground vs Precipitated Anatase; Report to Yale University Library; Yale University: New Haven, CT, 1975.
- Mackenzie, K. 3. D.; MaIling, P. 3. Trans. Br. Ceram. Soc. 1974, 73, 23-27.
- Towe, K. M. The Vinland Map Revisited: An Analysis of the McCrone Reports and an Evaluation of the Problem of the Map's Authenticity; Report to Yale University Library; Yale University: New Haven, CT, 1982.
- Olin, 3. S.; Towe, K. M. Abstracts, International Congress for the History of Cartography; Washington, DC, 1976; p 25.
- Cahill, T. A.; Schwab, R. N.; Kusko, B. H.; Ridred, R. A.; Moller, G.; Dutschke, D.; Wick, D. L.; Pooley, A. S. Further Elemental Analyses of the Vinland Map, the Tartar Relation, and the Speculum Historiale; Report to Yale University Beinecke Rare Book and Manuscript Library; Yale University: New Haven, CT, 1985.
- Clark, G. L. Applied X-rays, 4th ad.; McGraw-Hill: New York, 1955.
- King, H. P.; Alexander, L. E. X-ray Diffraction Procedures for Polycrystalline and Amorphous Materials, 2nd ed.; 3. Wiley & Sons: New York, 1974.
Biography:
Kenneth M. Towe was born on January 31, 1935, in Jacksonville, FL. He obtained an A.B. degree from Duke University in 1956, an M.Sc. degree from Brown University in 1959, and in 1961 a Ph.D. from the University of Illinois (Champaign Urbana) with a major in Geology and minor in Chemistry. He then spent one year at Illinois as Research Associate in the Electron Microscope Laboratory. Following completion of a two year postdoctoral position in the Division of Geological Sciences at the California Institute of Technology, Dr. Towe was employed in 1964 as a Research Geologist in the Department of Paleobiology at the Smithsonian Institution's National Museum of Natural History. Research interests include the mineralogy of clays and colloidal iron oxides, the study of invertebrate biomineralization, and research into the environmental and evolutionary history of early life on Earth. He retired as Senior Scientist in 1996.
[Note: Biography updated to be current as of 2003]
This article was published in the journal "Accounts of Chemical Research, 1990, 23." from the American Chemical Society in 1990. Received September 21, 1989 (Revised Manuscript Received December 18, 1989) Registry No. Anatase, 1317‑70‑0.
< Previous Next > |